This article provides an overview of the types of corrosion that occur and possible constructive measures to prevent or reduce corrosion.
In practice it is sometimes difficult to foresee the expected corrosion phenomena completely and precisely for the first product design. This can be remedied by the guidelines and regulations that may exist for the respective product, into which corresponding empirical values, such as additional values for the material thickness, have already been incorporated. If there are no such regulations, the following list is helpful for constructive measures against corrosion.
Types of corrosion
There are different types of corrosion, but what they have in common is that an electrolyte is usually required to start the corrosion. The following types can be distinguished:
- Uniform surface corrosion
- Peptic corrosion
- Pitting corrosion
- Crevice corrosion
Corrosion of contacting surfaces
- Contact corrosion
- Sediment corrosion
- Corrosion at phase interfaces
- Stress corrosion cracking
- Vibration corrosion cracking
- Corrosion induced by strain
- Erosion and cavitation corrosion
- Friction corrosion
- Intergranular corrosion
- Graphite corrosion
- Dezincification
The following describes the individual types of corrosion, their causes and the respective design measures against corrosion.
Corrosion of free surfaces
Uniform surface corrosion
Root cause
A metallic surface is exposed to moisture (weakly basic or acidic) and oxygen (normal contact with the atmosphere). These environmental conditions lead to material erosion over the entire surface due to corrosion.
Design countermeasures
- use stainless steel
- plan additional material thickness
- design to minimal surface area
- Allow areas to be ventilated to dry
- avoid the accumulation of liquids (e.g. plan inclined surfaces for drainage)
- avoid falling below the dew point (formation of condensate)
- use an anti-corrosive surface coating
- use cathodic protection
Peptic and pitting corrosion
Both peptic and pitting corrosion are locally increased free types of corrosion that are caused by inhomogeneities in the material. The design countermeasures apply as described above.
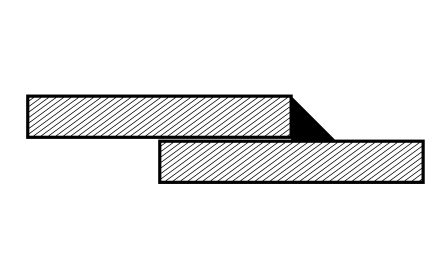
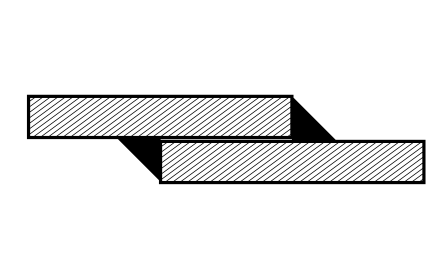
Crevice corrosion
Root cause
Electrolytes can accumulate in crevices and accelerate corrosion. In addition, the accumulation of electrolytes leads to a lack of oxygen, which can also cause damage to corrosion- and acid-resistant steels.
Design countermeasures
- weld overlapping material on both sides
- apply sealing runs to weld seams
- do not use interrupted weld seams
- seal the gap
- increase the gap to allow ventilation
Corrosion of contacting surfaces
Contact corrosion
Root cause
Use of metals with different potentials (electrochemical series) with the simultaneous presence of an electrolyte.
Design countermeasures
- use materials with a lower potential difference
- isolate the two materials
- avoid electrolyte
- use of sacrificial anodes
Sediment corrosion
Root cause
The origin of sediment corrosion is also in the electrochemical series, i.e. the potential difference. Sediments can accumulate during production (e.g. extraneous rust) as well as during operation due to abrasion or other effects and lead to corrosion.
Design countermeasures
- avoidance of deposit products/sediment through filtering or cleaning
- in the case of machine parts with flow: avoiding "dead" flow zones in which particles can settle
Corrosion at phase interfaces
Root cause
In the transition area between, for example, the liquid phase and the vapor phase of a medium, increased corrosion can occur. This can occur both with stationary liquids and with "changing" media, e.g. in the transition zone from water to water vapor in a heat exchanger.
Design countermeasures
- minimize the component area in the area of the phase interface
- slow down the phase change (with flowing media)
- vary fill levels
Corrosion from stress
Stress corrosion cracking
Root cause
There are materials that are susceptible to stress corrosion cracking. These are:
- unalloyed carbon steels
- austenitic steels
- Brass
- Magnesium alloys
- Aluminum alloys
- Titanium alloys
Stress corrosion cracking can be triggered by external tensile stresses or the internal stress state of a component.
Design countermeasures
- avoid susceptible materials
- lowering the tensile stress (in the area of the surface)
- application of compressive stress
- stress relief heat treatment
- avoid electrolytes
- use cathodic coatings
Vibration corrosion cracking
Root cause
The vibration corrosion cracking is difficult to distinguish from fatigue failure because it shows almost the same characteristics.
Design countermeasures
- avoid alternating stress
- avoid grooves
- avoid electrolytes
- Introduce compressive prestress into the surface of the component (e.g. by shot peening)
- use surface coating
Strain corrosion
Root cause
The expansion or compression of a component can damage a protective surface layer or surface coating.
Design countermeasures
- reduce strain or compression
Erosion
Root cause
Surface damage caused by particles contained in a flowing fluid.
Design countermeasures
- filtering the particles
- decrease the flow velocity
- redirecting the flow
- apply additional surface layers, such as build-up welds, nickel layers, hard chrome or stellite
Cavitation
Root cause
Cavitation leads to local overstressing of the surface, so that corrosion is favored at these points.
Design countermeasures
- decrease the flow velocity
- apply additional surface layers, such as build-up welds, nickel layers, hard chrome or stellite
Friction corrosion
Root cause
Friction from moving, adjoining components leads to both abrasion and higher surface stress due to surface pressure.
Design countermeasures
- use of interchangeable friction materials
- use of lubricants
- avoid the motion
- avoid the contact
Selective corrosion
Root cause
Selective corrosion is material corrosion on the crystal lattice level. The material can be more susceptible to corrosion at the grain boundaries, so that e.g. in cast iron, the iron components dissolve (spongiosis or graphite corrosion).
Intracrystalline corrosion particularly affects austenitic CrNi steels after unsuitable heat treatment.
Design countermeasures
- apply suitable heat treatment measures for the materials used

